Introduction to non-specific factors of the organism resistance
1. Types of organism resistance barriers
Innate immunity comprises four types of defensive barriers: anatomic, physiologic, phagocytic, and inflammatory (Table 1-2).

2.The skin and the mucosal surfaces provide protective barriers against infection
Physical and anatomic barriers that tend to prevent the entry of pathogens are an organism’s first line of defense against infection. The skin and the surface of mucous membranes are included in this category because they are effective barriers to the entry of most microorganisms. The skin consists of two distinct layers: a thinner outer layer – the epidermis – and a thicker layer – the dermis. The epidermis contains several layers of tightly packed epithelial cells. The outer epidermal layer consists of dead cells and is filled with a waterproofing protein called keratin. The dermis, which is composed of connective tissue, contains blood vessels, hair follicles, sebaceous glands, and sweat glands. The sebaceous glands are associated with the hair follicles and produce an oily secretion called sebum. Sebum consists of lactic acid and fatty acids, which maintain the pH of the skin between 3 and 5; this pH inhibits the growth of most microorganisms. A few bacteria that metabolize sebum live as commensals on the skin and sometimes cause a severe form of acne (угорь, прыщ). One acne drug, isotretinoin (Accutane), is a vitamin A derivative that prevents the formation of sebum.
Breaks in the skin resulting from scratches (царапины), wounds, or abrasion (ссадина) are obvious routes of infection. The skin may also be penetrated by biting insects (e.g., mosquitoes, mites, ticks, fleas, and sandflies - комары, клещи, клещи, блохи, и москиты); if these harbor pathogenic organisms, they can introduce the pathogen into the body as they feed. The protozoan that causes malaria, for example, is deposited in humans by mosquitoes when they take a blood meal. Similarly, bubonic plague (бубонная чума) is spread by the bite of fleas, and Lyme disease (tick fever or borreliosis) is spread by the bite of ticks, it is an infectious disease caused by at least three species of genus Borrelia bacteria.
The conjunctivae and the alimentary, respiratory, and urogenital tracts are lined by mucous membranes, not by the dry, protective skin that covers the exterior of the body. These membranes consist of an outer epithelial layer and an underlying layer of connective tissue. Although many pathogens enter the body by binding to and penetrating mucous membranes, a number of nonspecific defense mechanisms tend to prevent this entry. For example, saliva, tears, and mucous secretions act to wash away potential invaders (захватчики) and also contain antibacterial or antiviral substances. The viscous fluid called mucus, which is secreted by epithelial cells of mucous membranes, entraps (улавливает) foreign microorganisms. In the lower respiratory tract, the mucous membrane is covered by cilia, hairlike protrusions (выпячивание) of the epithelial-cell membranes. The synchronous movement of cilia propels mucus-entrapped microorganisms from these tracts. In addition, nonpathogenic organisms tend to colonize the epithelial cells of mucosal surfaces. These normal flora generally outcompete pathogens for attachment sites on the epithelial cell surface and for necessary nutrients.
Some organisms have evolved ways of escaping these defense mechanisms and thus are able to invade the body through mucous membranes. For example, influenza virus (the agent that causes flu) has a surface molecule that enables it to attach firmly (крепко) to cells in mucous membranes of the respiratory tract, preventing the virus from being swept out by the ciliated epithelial cells. Similarly, the organism that causes gonorrhea has surface projections that allow it to bind to epithelial cells in the mucous membrane of the urogenital tract. Adherence of bacteria to mucous membranes is due to interactions between hairlike protrusions on a bacterium, called fimbriae or pili, and certain glycoproteins or glycolipids that are expressed only by epithelial cells of the mucous membrane of particular tissues. For this reason, some tissues are susceptible to bacterial invasion, whereas others are not.
3. Physiologic barriers to infection include general conditions and specific molecules
The physiologic barriers that contribute to innate immunity include temperature, pH, and various soluble and cellassociated molecules. Many species are not susceptible to certain diseases simply because their normal body temperature inhibits growth of the pathogens. Chickens, for example, have innate immunity to anthrax because their high body temperature inhibits the growth of the bacteria. Gastric acidity is an innate physiologic barrier to infection because very few ingested microorganisms can survive the low pH of the stomach contents. One reason newborns are susceptible to some diseases that do not afflict adults is that their stomach contents are less acid than those of adults.
A variety of soluble factors contribute to innate immunity, among them the soluble proteins lysozyme, interferon, and complement. Lysozyme, a hydrolytic enzyme found in mucous secretions and in tears, is able to cleave the peptidoglycan layer of the bacterial cell wall. Interferon comprises a group of proteins produced by virus-infected cells. Among the many functions of the interferons is the ability to bind to nearby cells and induce a generalized antiviral state. Complement is a group of serum proteins that circulate in an inactive state. A variety of specific and nonspecific immunologic mechanisms can convert the inactive forms of complement proteins into an active state with the ability to damage the membranes of pathogenic organisms, either destroying the pathogens or facilitating their clearance. Complement may function as an effector system that is triggered by binding of antibodies to certain cell surfaces, or it may be activated by reactions between complement molecules and certain components of microbial cell walls. Reactions between complement molecules or fragments of complement molecules and cellular receptors trigger activation of cells of the innate or adaptive immune systems. Recent studies on collectins indicate that these surfactant proteins may kill certain bacteria directly by disrupting their lipid membranes or, alternatively, by aggregating the bacteria to enhance their susceptibility to phagocytosis.
Many of the molecules involved in innate immunity have the property of pattern recognition, the ability to recognize a given class of molecules. Because there are certain types of molecules that are unique to microbes and never found in multicellular organisms, the ability to immediately recognize and combat invaders displaying such molecules is a strong feature of innate immunity. Molecules with pattern recognition ability may be soluble, like lysozyme and the complement components described above, or they may be cell-associated receptors.
Among the class of receptors designated the toll-like receptors (TLRs), TLR2 recognizes the lipopolysaccharide (LPS) found on Gram-negative bacteria. It has long been recognized that systemic exposure of mammals to relatively small quantities of purified LPS leads to an acute inflammatory response (see below). The mechanism for this response is via a TLR on macrophages that recognizes LPS and elicits a variety of molecules in the inflammatory response upon exposure. When the TLR is exposed to the LPS upon local invasion by a Gram-negative bacterium, the contained response results in elimination of the bacterial challenge.
4. Cells that ingest and destroy pathogens make up a phagocytic barrier to infection
Another important innate defense mechanism is the ingestion of extracellular particulate material by phagocytosis. Phagocytosis is one type of endocytosis, the general term for the uptake by a cell of material from its environment. In phagocytosis, a cell’s plasma membrane expands around the particulate material, which may include whole pathogenic microorganisms, to form large vesicles called phagosomes (Figure 1). Most phagocytosis is conducted by specialized cells, such as blood monocytes, neutrophils, and tissue macrophages. Most cell types are capable of other forms of endocytosis, such as receptor-mediated endocytosis, in which extracellular molecules are internalized after binding by specific cellular receptors, and pinocytosis, the process by which cells take up fluid from the surrounding medium along with any molecules contained in it.
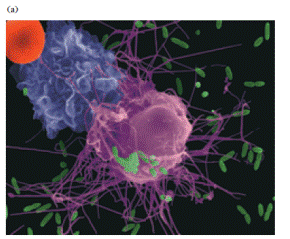
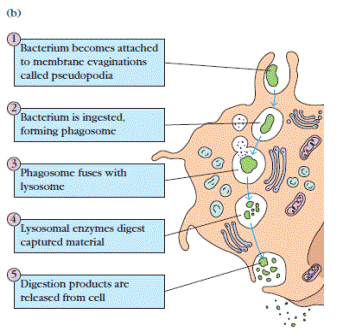
Figure 1. (a) Electronmicrograph of macrophage (pink) attacking Escherichia coli (green). The bacteria are phagocytized as described in part b and breakdown products secreted. The monocyte (purple) has been recruited to the vicinity of the encounter by soluble factors secreted by the macrophage. The red sphere is an erythrocyte. (b) Schematic diagram of the steps in phagocytosis of a bacterium. [Part a, Dennis Kunkel Microscopy, Inc./Dennis Kunkel.]
5. Inflammation represents a complex sequence of events that stimulates immune responses
Tissue damage caused by a wound or by an invading pathogenic microorganism induces a complex sequence of events collectively known as the inflammatory response. As described above, a molecular component of a microbe, such as LPS, may trigger an inflammatory response via interaction with cell surface receptors. The end result of inflammation may be the marshalling of a specific immune response to the invasion or clearance of the invader by components of the innate immune system. Many of the classic features of the inflammatory response were described as early as 1600 BC, in Egyptian papyrus writings. In the first century AD, the Roman physician Celsus described the “four cardinal signs of inflammation” as rubor (redness), tumor (swelling), calor (heat), and dolor (pain). In the second century AD, another physician, Galen, added a fifth sign: functio laesa (loss of function). The cardinal signs of inflammation reflect the three major events of an inflammatory response (Figure 2):
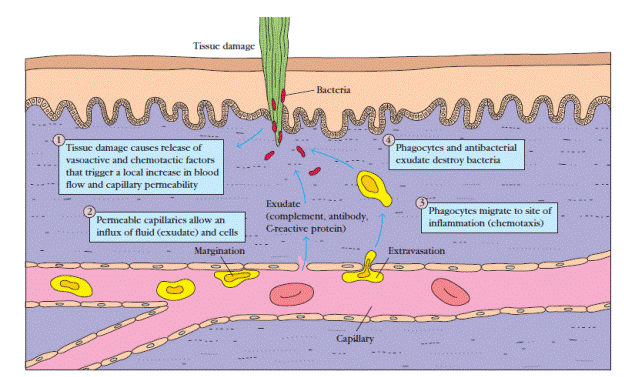
Figure 2 Major events in the inflammatory response. A bacterial infection causes tissue damage with release of various vasoactive and chemotactic factors. These factors induce increased blood flow to the area, increased capillary permeability, and an influx of white blood cells, including phagocytes and lymphocytes, from the blood into the tissues. The serum proteins contained in the exudate have antibacterial properties, and the phagocytes begin to engulf the bacteria.
1. Vasodilation – an increase in the diameter of blood vessels – of nearby capillaries occurs as the vessels that carry blood away from the affected area constrict, resulting in engorgement of the capillary network. The engorged capillaries are responsible for tissue redness (erythema) and an increase in tissue temperature.
2. An increase in capillary permeability facilitates an influx of fluid and cells from the engorged capillaries into the tissue. The fluid that accumulates (exudate) has a much higher protein content than fluid normally released from the vasculature. Accumulation of exudate contributes to tissue swelling (edema).
3. Influx of phagocytes from the capillaries into the tissues is facilitated by the increased permeability of the capillaries. The emigration of phagocytes is a multistep process that includes adherence of the cells to the endothelial wall of the blood vessels (margination), followed by their emigration between the capillaryendothelial cells into the tissue (diapedesis or extravasation), and, finally, their migration through the tissue to the site of the invasion (chemotaxis). As phagocytic cells accumulate at the site and begin to phagocytose bacteria, they release lytic enzymes, which can damage nearby healthy cells. The accumulation of dead cells, digested material, and fluid forms a substance called pus.
The events in the inflammatory response are initiated by a complex series of events involving a variety of chemical mediators whose interactions are only partly understood. Some of these mediators are derived from invading microorganisms, some are released from damaged cells in response to tissue injury, some are generated by several plasma enzyme systems, and some are products of various white blood cells participating in the inflammatory response.
Among the chemical mediators released in response to tissue damage are various serum proteins called acute-phase proteins. The concentrations of these proteins increase dramatically in tissue-damaging infections. C-reactive protein is a major acute-phase protein produced by the liver in response to tissue damage. Its name derives from its patternrecognition activity: C-reactive protein binds to the C-polysaccharide cell-wall component found on a variety of bacteria and fungi. This binding activates the complement system, resulting in increased clearance of the pathogen either by complement-mediated lysis or by a complementmediated increase in phagocytosis.
One of the principal mediators of the inflammatory response is histamine, a chemical released by a variety of cells in response to tissue injury. Histamine binds to receptors on nearby capillaries and venules, causing vasodilation and increased permeability. Another important group of inflammatory mediators, small peptides called kinins, are normally present in blood plasma in an inactive form.Tissue injury activates these peptides, which then cause vasodilation and increased permeability of capillaries. A particular kinin, called bradykinin, also stimulates pain receptors in the skin. This effect probably serves a protective role, because pain normally causes an individual to protect the injured area.
Vasodilation and the increase in capillary permeability in an injured tissue also enable enzymes of the blood-clotting system to enter the tissue. These enzymes activate an enzyme cascade that results in the deposition of insoluble strands of fibrin, which is the main component of a blood clot. The fibrin strands wall off the injured area from the rest of the body and serve to prevent the spread of infection.
Once the inflammatory response has subsided and most of the debris has been cleared away by phagocytic cells, tissue repair and regeneration of new tissue begins. Capillaries grow into the fibrin of a blood clot. New connective tissue cells, called fibroblasts, replace the fibrin as the clot dissolves. As fibroblasts and capillaries accumulate, scar tissue forms.
Дата добавления: 2016-07-18; просмотров: 2796;











